
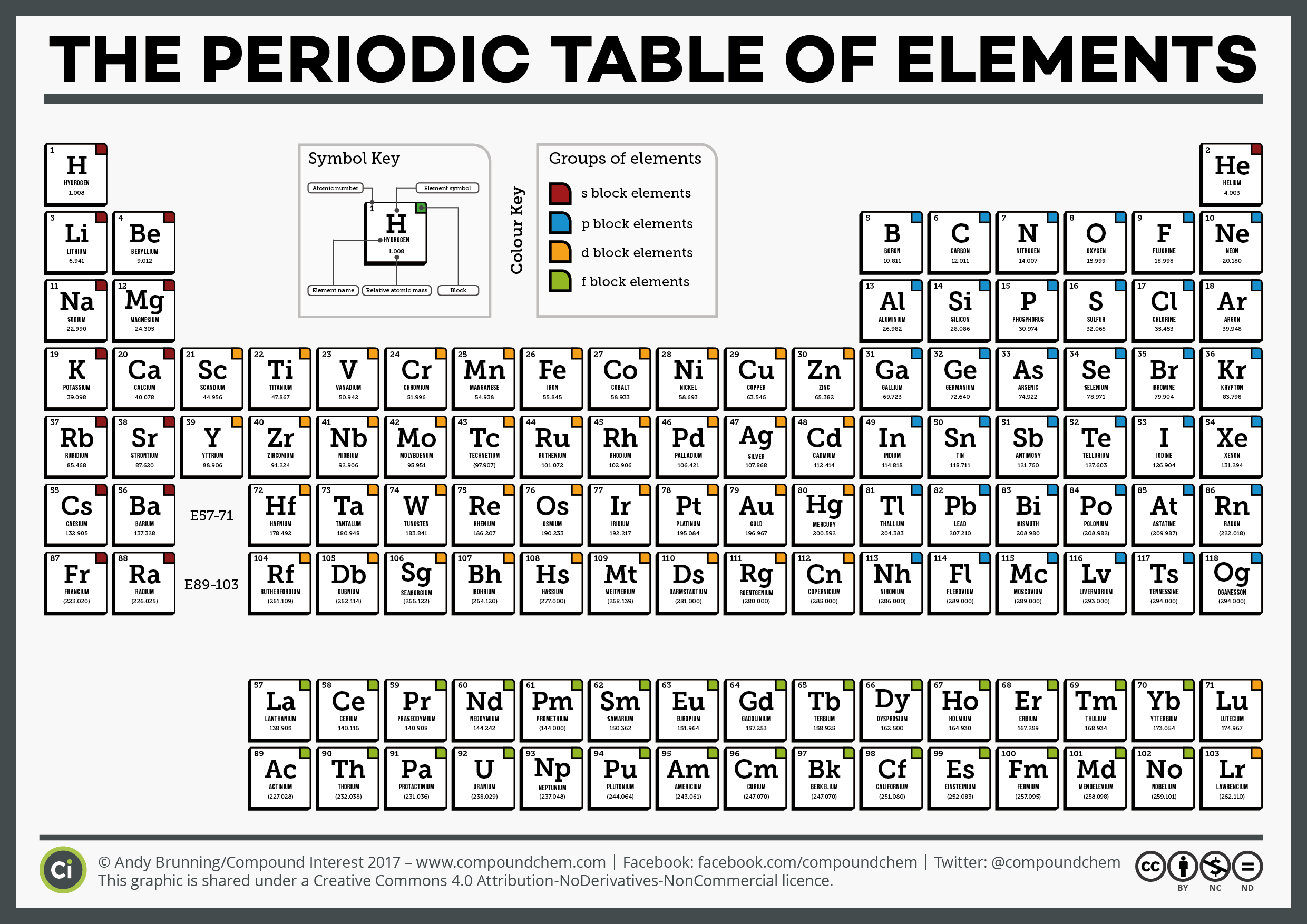
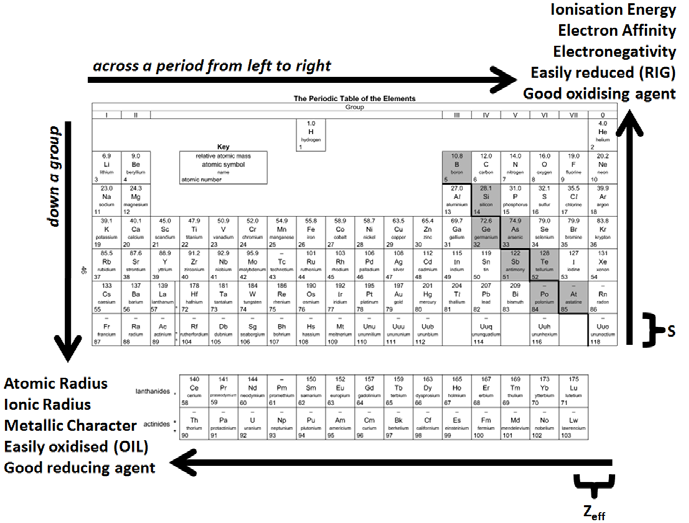
These electrons screen or shield the outer electrons from the nuclear charge. The periodic table and periodic trends might seem overwhelming, but with a little review with MCAT practice questions, you’ll be able to understand their complexities. This is because of the screening effect of the filled inner electron levels. It is important to note that Fluorine, which sits on the top right corner of the periodic table, is the most electronegative element. The greater attraction between the increased number of protons (increased nuclear charge) and electrons, pulls the electrons closer together, hence the smaller size.Īs you move down a group in the periodic table, the covalent radius increases. The covalent radius (a measure of how large individual atoms are) shows different trends if you are moving across a period or down a group.Ī comparison of the relative covalent radii of atoms is shown in the diagram below.Īcross a period from left to right, the covalent radius decreases.Īs you move from left to right across the periodic table, atoms have more electrons in their outer energy level and more protons in their nucleus. Meyer formed his periodic law based on the atomic volume or molar volume, which is the atomic mass divided by the density in solid form.Patterns and trends in the periodic tableĬhemists observe patterns in different properties of elements as they are arranged in the periodic table. They both arranged the elements by their mass and proposed that certain properties periodically reoccur. Mendeleev created the first periodic table and was shortly followed by Meyer.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
